REGENERA ACTIVA
A certified autologous micrografting protocol with tissue regeneration potential for the treatment of hair loss.

80+ STUDIES

CE CERTIFIED

60+ COUNTRIES
*For Healthcare Professionals (HCP)
SCROLL
COMPARE THE FACTS
Regenera Activa EVO protocol versus alternatives


Clinical evidence is the foundation of informed medical decisions.
Choose protocols backed by rigorous scientific validation.
Clinical evidence is the foundation of informed medical decisions.
Choose protocols backed by rigorous scientific validation.
TRUSTED BY PHYSICIANS WORLDWIDE
Global leaders in aesthetic medicine share their experience



PROTOCOL INTEGRITY
Three pillars of clinical excellence and patient safety

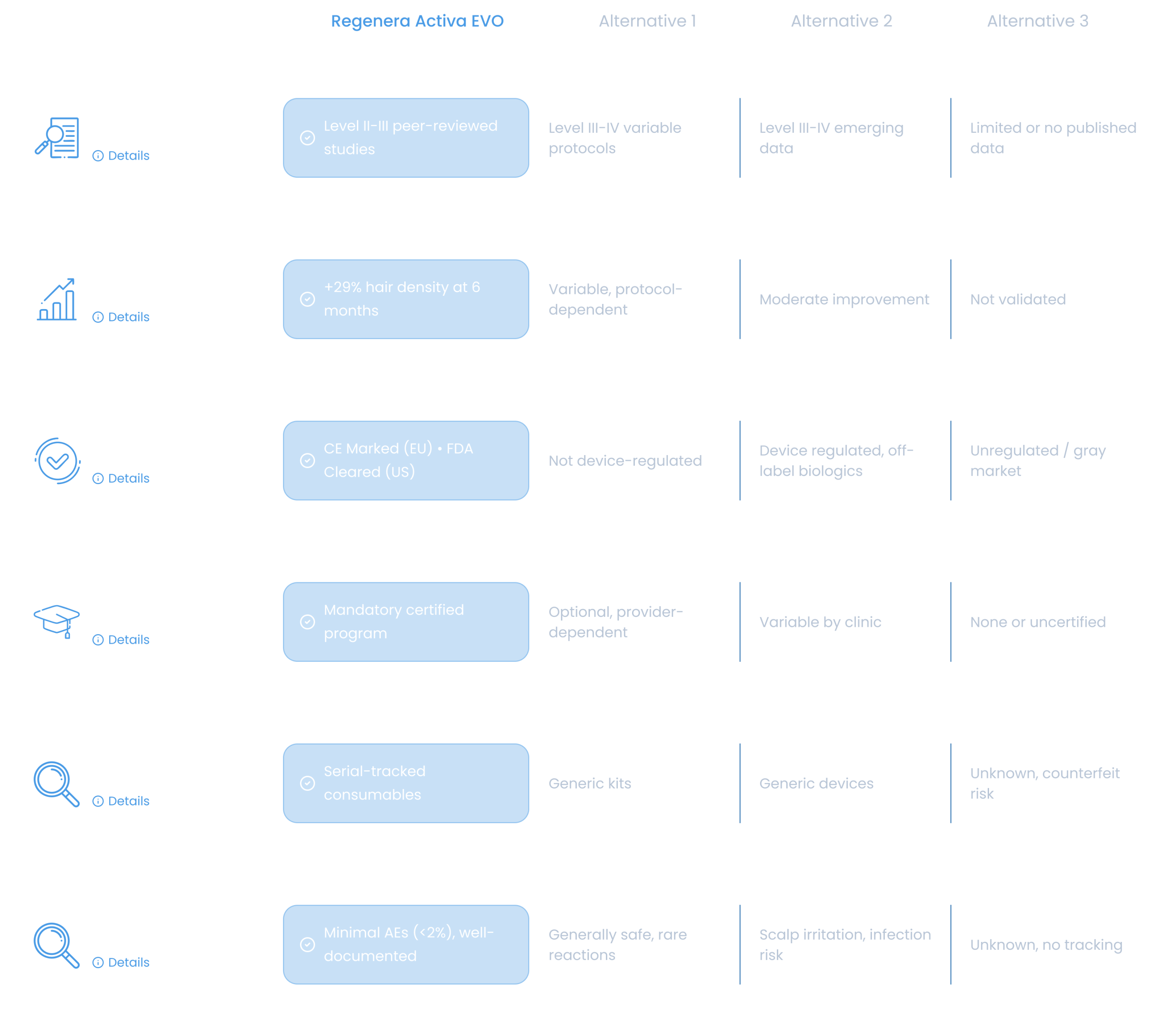
ORIGINAL DEVICE & PROTOCOL
Patented Regenera Activa EVO medical device with CE marking. Authentic equipment ensures consistent processing and optimal cell viability across all treatments.

CERTIFIED TRAINING
Mandatory physician certification program. Comprehensive training ensures protocol adherence and optimal patient outcomes through standardized techniques.

AUTHENTIC CONSUMABLES
Lot-tracked sterile kits with holographic seals. Traceable supply chain prevents counterfeit products and ensures complete safety compliance.
REGENERATIVE TREATMENTS COMPARISON
Regenera Activa EVO in the landscape of regenerative treatments
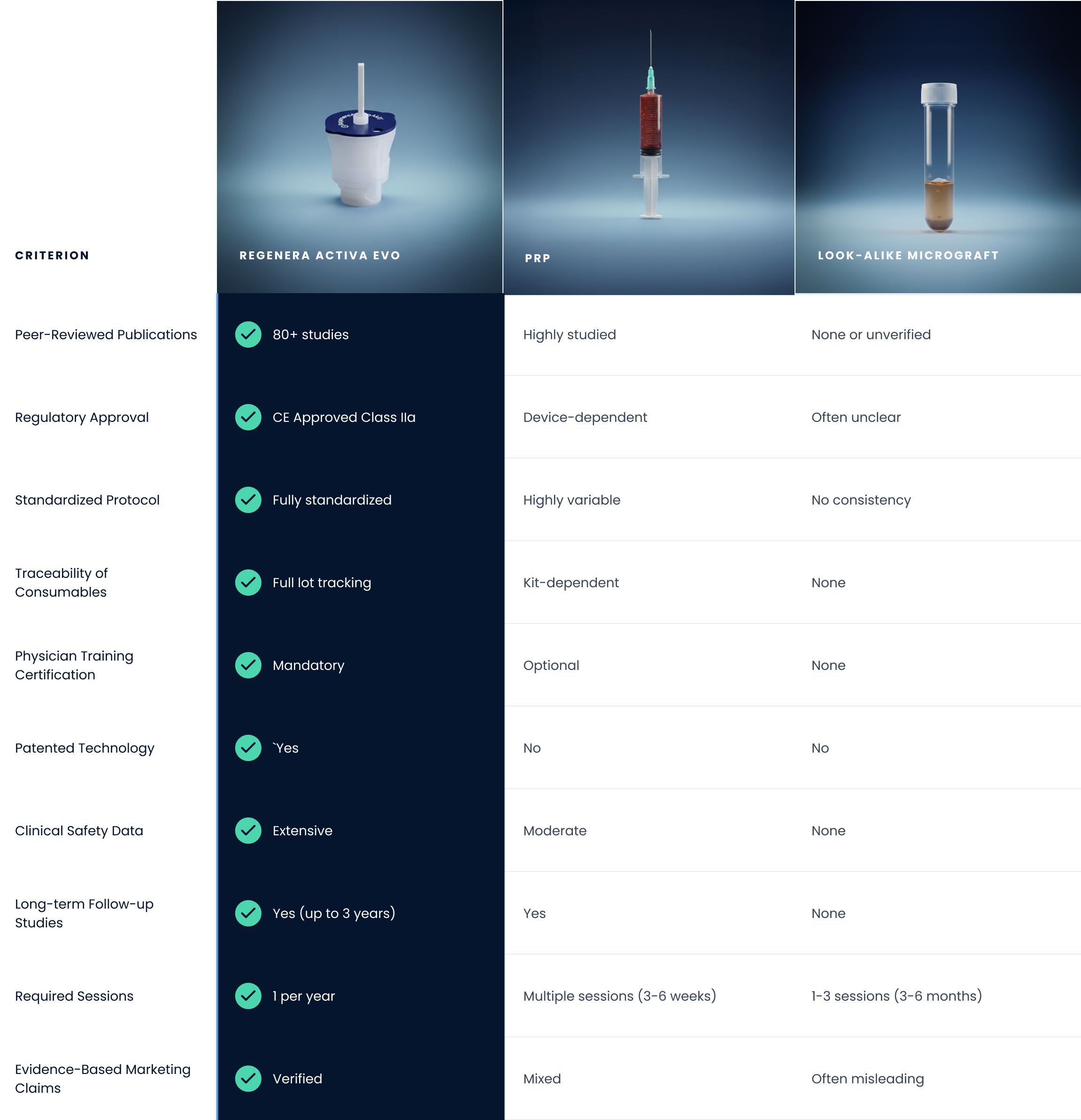
| CRITERION |  |
 |
 |
|---|---|---|---|
| Peer-Reviewed Publications |  80+ studies 80+ studies |
Highly studied | None or unverified |
| Regulatory Approval |  CE Approved Class IIa CE Approved Class IIa |
Device-dependent | Often unclear |
| Standardized Protocol |  Fully standardized Fully standardized |
Highly variable | No consistency |
| Traceability of Consumables |  Full lot tracking Full lot tracking |
Kit-dependent | None |
| Physician Training Certification |  Mandatory Mandatory |
Optional | None |
| Patented Technology |  Yes Yes |
No | No |
| Clinical Safety Data |  Extensive Extensive |
Moderate | None |
| Long-term Follow-up Studies |  Yes (Up to 3 years) Yes (Up to 3 years) |
Yes | None |
| Evidence-Based Marketing Claims |  Verified Verified |
Mixed | Often misleading |
| Required Sessions |  1 per year 1 per year |
Multiple sessions (3-6 weeks) | 1-3 sessions (3-6 months) |
Regenera Activa is the only proven autologous and homologous micrografting technology for hair loss treatments.
+30%
Avg. Hair Density Increase*
CE Approved + FDA Listed
Cleared & Certified
30
Minutes Per Session
0%
Adverse Events**
+30%
Avg. Hair Density Increase*
CE Approved + FDA Listed
Cleared & Certified
30
Minutes Per Session
0%
Adverse Events**
* Ruiz, R.G.; Rosell, J.M.C.; Ceccarelli, G.; De Sio, C.; De Angelis, G.; Pinto, H.; Astarita, C.; Graziano, A. Progenitor-cell-enriched micrografts as a novel option for the management of androgenetic alopecia. J. Cell Physiol. 2019, 235, 4587–4593.
**Directly associated with the final AMT Suspension. If there are some secondary effects they would be related to the technique (administration method)
COMPARE THE FACTS
Side-by-side analysis of certified protocols versus alternatives
Want the Complete Clinical Data Package?
Download detailed study summaries, regulatory documents, and training materials
READY TO ELEVATE YOUR
HAIR REGENERATION PRACTICE?
Connect with our Medical Affairs team to learn how certified protocols ensure patient safety,
optimal outcomes, and complete regulatory compliance.

Talk to a specialist
Connect with our Medical Affairs team for detailed Questions & Answers.
No obligations • For Healthcare Professionals only • HIPAA compliant
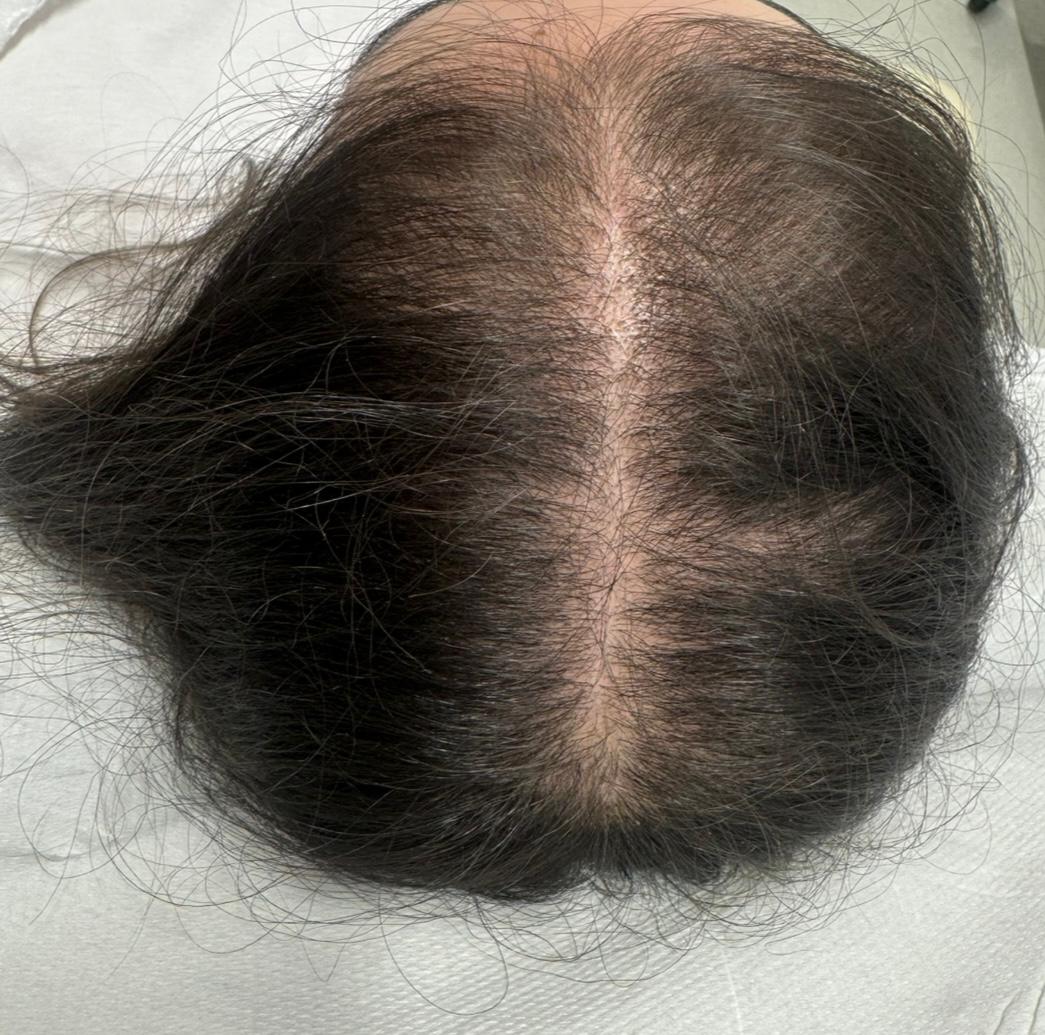
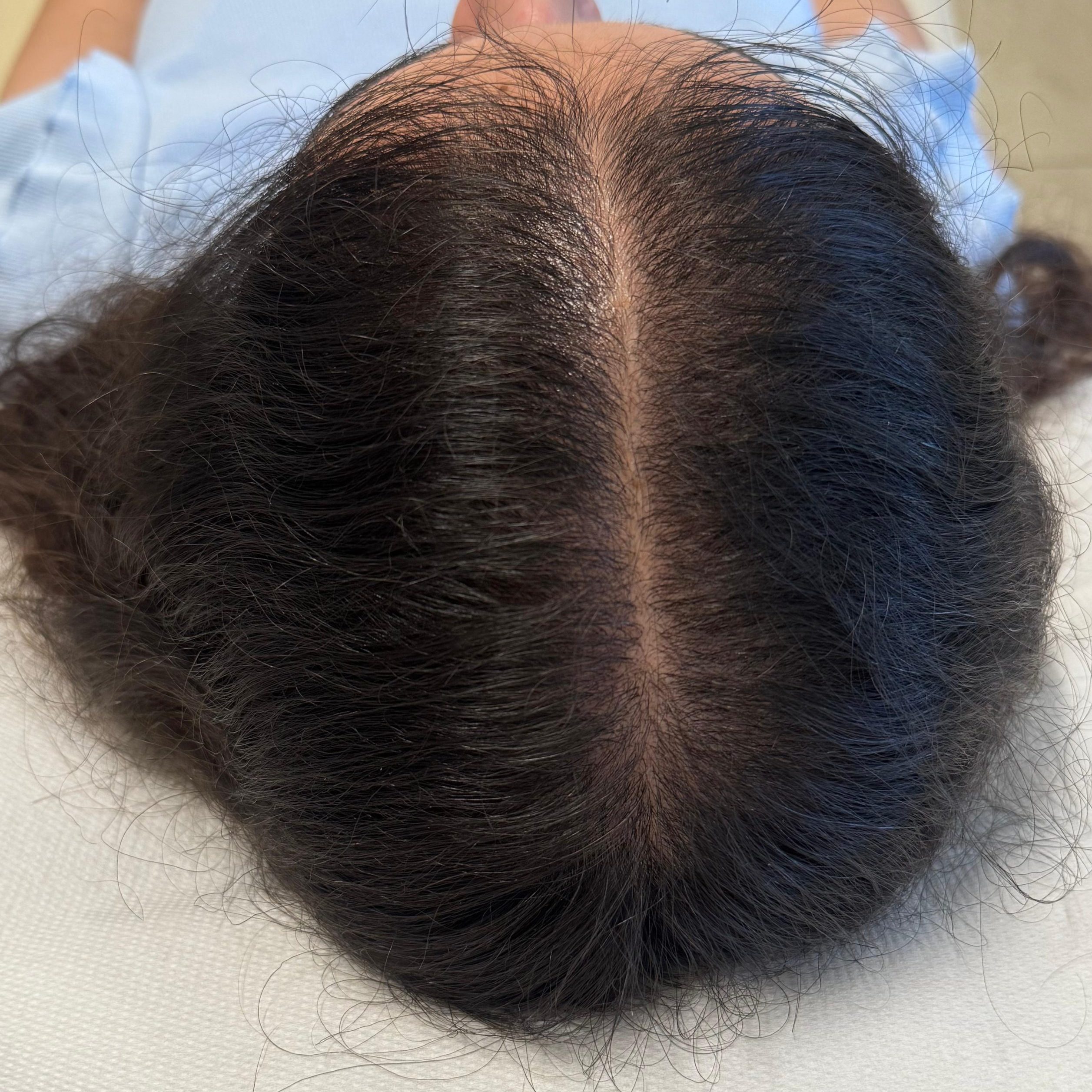
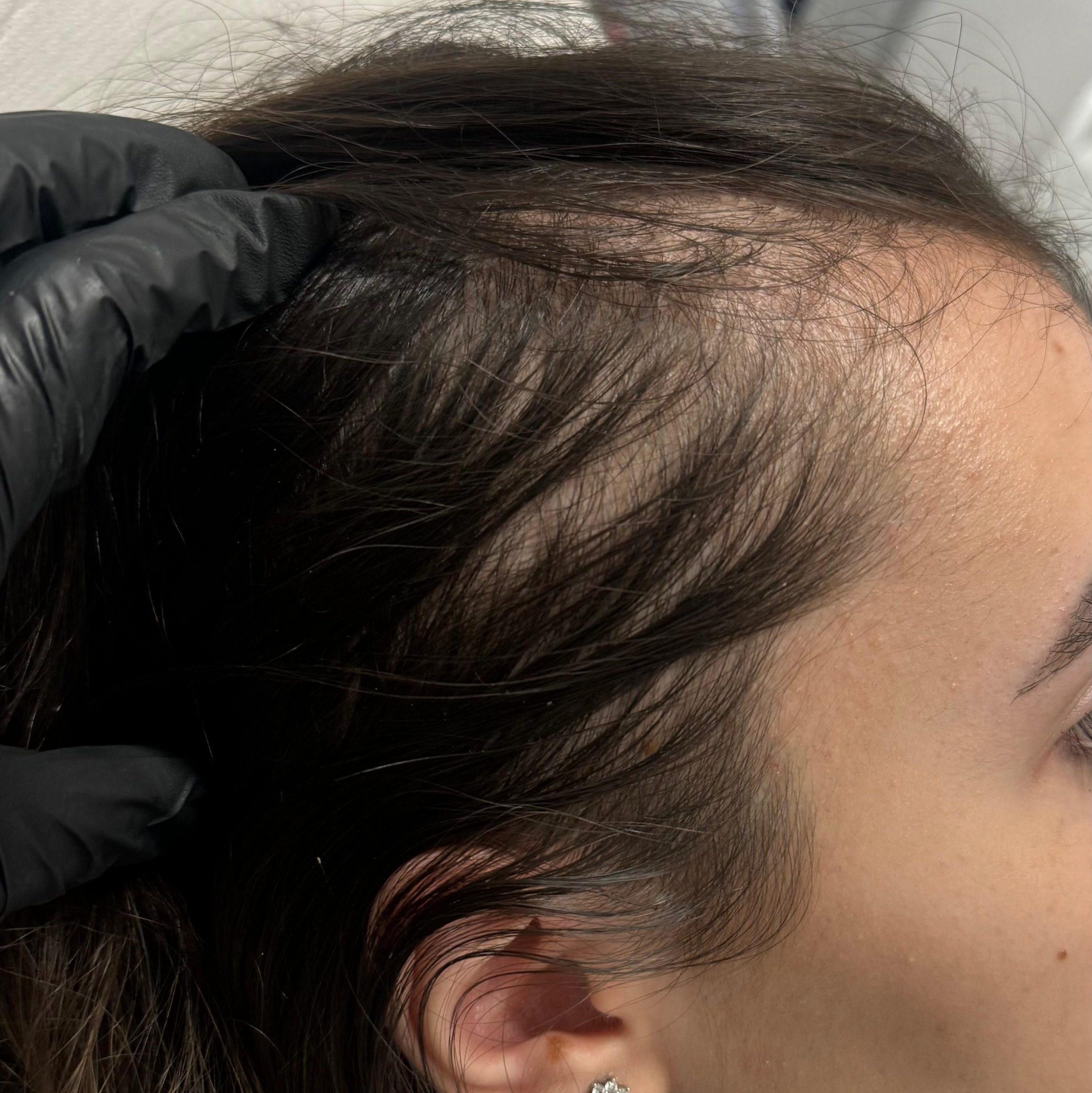
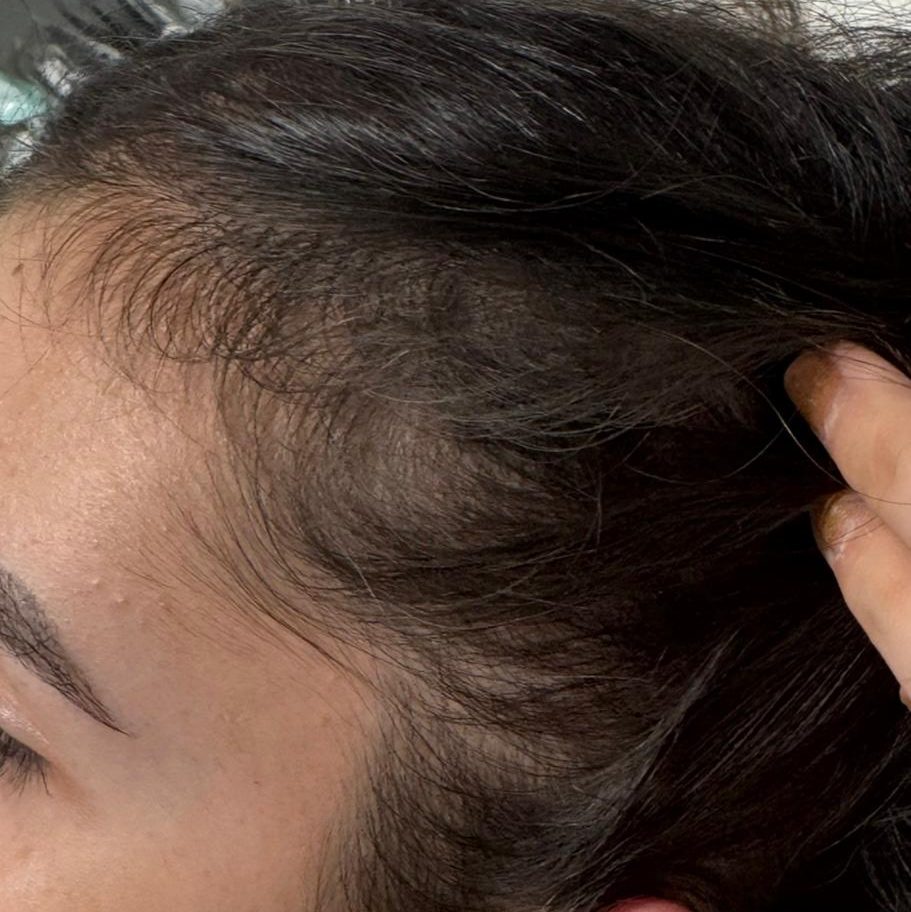
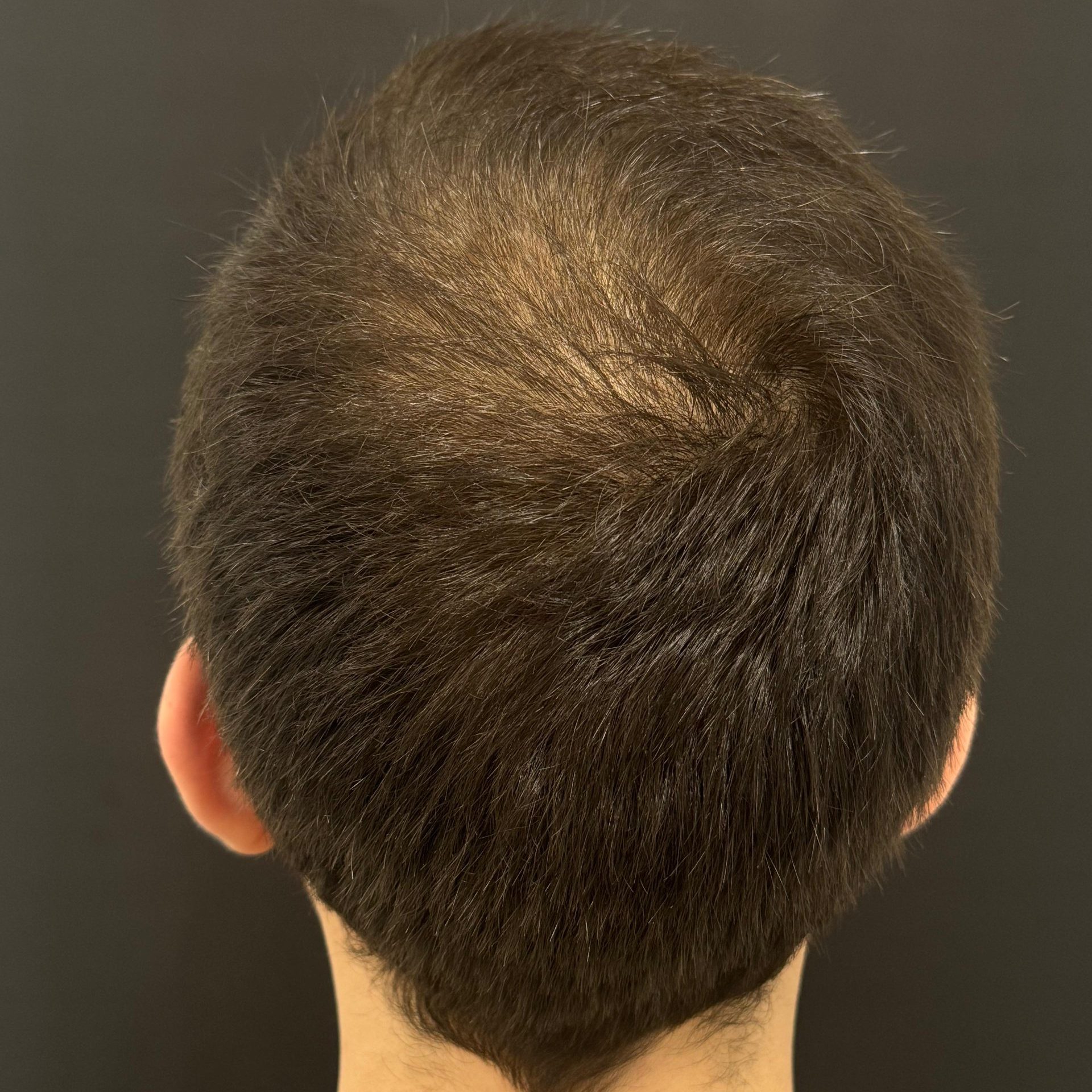
BEFORE AND AFTER WITH REGENERA ACTIVA EVO
With Regenera Activa EVO patients may start seeing cosmetic results after 3 months.
FREQUENTLY ASKED QUESTIONS
Common questions about protocol integrity and verification.
01. What makes Regenera Activa EVO different from PRP treatments?
Regenera Activa EVO uses autologous micrografts containing progenitor cells from scalp tissue, whereas PRP uses blood-derived platelets. The micrograft approach has demonstrated superior and more consistent outcomes in comparative studies, with a mean hair density increase of 29% at 6 months.
02. Why was EVO developed, and what makes it unique?
As leaders in the field of Hair Regeneration, we are committed to constant research and development. EVO was developed after 10 years of innovation and scientific validation to improve the quality and quantity of micrografts in our solution. The changes in materials and technology ensure consistent pressure, precise rotation, and better tissue disaggregation, making the procedure safer, faster, and more efficient.
03. How does EVO achieve 70 billion exosomes in one session?
EVO’s improved disaggregation process increases the quantity of micrografts, which enhances all their components, including exosomes, progenitor cells, and growth factors.
04. How do the mechanical improvements in EVO enhance results?
EVO ensures better tissue processing with homogeneous pressure and precise rotation. This leads to up to 50% more micrografts, improved cell viability (+90%), and a more effective regenerative solution.